Renowned Speakers

Yavuz S. Silay
MaQasid Angel Investors Network Turkey Turkey

Hongbin Wang
California Northstate University USA

Wafa T. Al-Jamal
University of East Anglia UK

Maysoon A Abujarad Alhuwitat
Amman Arab University Jordan

Paul Fisher
Inovio Pharmaceuticals Inc USA

Reveng Abdulkareem
University of Duhok Iraq

Arwyn T Jones
Cardiff University Sweden

Andreas Bernkop-Schnürch
University of Innsbruck Austria
Recommended Global Pharmaceutical Sciences Webinars & Conferences
Europe & UK
Asia Pacific & Middle East
Canada
Pharma Research 2024
Pharmaceutical Research
May 16 - May 17, 2024 | Rome, Italy
Conference Series LLC Ltd welcomes you to attend the Pharmaceutical Research & Innovations in Pharma Industry Conference to be held in Rome, Italy on May 16-17, 2024. The theme for the conference this year is "Innovations in Pharma Research: Pioneering Health for a Sustainable Future"
Details of Pharma Research 2024 Conferences in Austria
|
Conference Name |
Place |
Date |
|
Rome, Italy |
May 16-17, 2024 |
About Conference
Global Pharmaceutical and Pharma Industry Conference scheduled during May 16-17 at Rome, Italy. The Conference emphasizes the theme “Exploring Innovative Developments in Pharmaceutical & Pharma Industry”. It covers a wide range of important sessions. Pharma Conference 2024 is a two-day program which includes thought inspiring Keynote Presentations, Plenary Talks, Symposiums, Special Sessions and Career Development Programs.
The conference will also witness professionals gathering from Academic, Pharmaceutical industry Research and Development wings, API: Active Pharmaceutical Ingredients industry, Drug discovery laboratories, Formulations and NDDS domains. Also executives and eminent research personalities from the departments pharmaceutical Chemistry: Quality control and Quality assurance, Clinical trials and Pharmacovigilance, Pharmaceutical Manufacturing, Pharamacognosy, Biopharmaceutics, Drug Discovery, Design & Development and Pharmacoeconomics shall be an integral part of this conference.
Global Pharmaceutical and Pharma Industry Conference provides an optimistic platform for exchanging ideas and helps the attendees to get updated about the advancements and innovations in Pharma Industry. Take a chance and make it count in your Professional life, Attend the Pharma Conference 2024 to network with your peers, exchange expertise and experiences, and arm yourself with the latest information to take your department to the next level.
Importance & Scope:
Pharma Conference 2024 is a best platform to present and discuss current aspect in drug research and development. Pharmachemistry is the study of interaction of chemical with living systems. It is important to discuss the pharmacological basis of therapeutics in order to maximize the benefits and minimize the risks of drugs to recipients. Hence it will be the best platform for scientists, researchers, PhD Scholars, Graduates and Post Graduates involved with drug development.
Global Pharmaceutical Chemicals Market: Key Research Aspects
This report analyzes and forecasts the market for pharmaceutical at the global and regional level. The market has been forecast based on volume (Tons) and revenue (US$ Mn) from 2017 to 2025, considering 2016 as the base year. The study includes drivers and restraints of the global pharmaceutical market. It also covers impact of these drivers and restraints on demand for pharmaceutical during the forecast period. The report also highlights opportunities in the pharmaceutical chemicals market at the global and regional level.
The global pharmaceutical market has been segmented as follows:
- Pharmaceutical Market: Type Analysis
- Solvents
- Reagents/Catalysts
- KSMs/intermediates
- Building blocks for APIs/Advanced intermediates
- Pharmaceutical chemicals Market: Drug Type Analysis
- OTC
- Generic
- Super generic
- Proprietary
Session/Tracks
Track 1: Pharmaceutical Sciences:
Pharmaceutical sciences combine a broad range of scientific disciplines that are involved with the design, action, delivery, disposition and use of drugs. This field draws on numerous regions of the essential and connected sciences, for example, science, science, the study of disease transmission, measurements, chemo metrics, arithmetic, material science and compound building and applies their standards to the investigation of medications.
Pharmaceutical formulations:
- Bio pharmaceutics
- Pre formulation studies
- Pharmaceutical biotechnology
- Routes of drug delivery
- Pharmaceutical materials
Track 2: Technology in Pharma Sciences:
Healthcare information technology is the area of information technology system involving the design, development, creation, use and maintenance of information system approaches to improve public health, medical care, lower cost, increase efficiency, reduce errors and improve patient satisfaction for the healthcare industry. These include uses of data analytics, artificial intelligence, cognitive computing and many others to effectively manage patient health management programs, development of new therapies and drugs, efficient aggregation and analysis of patient information.
Track 3: Pharmaceutical Manufacturing, Scale Up and Tech transfer:
The biopharmaceutical sector in US enjoys 17% of all domestic R&D funded by the country and in return gave $2.5 trillion cumulative addition in economic output in 2016. Companies engaged in the brand name pharmaceutical manufacturing have grappled in recent years owing to the largest waves of drug patent expirations in history. As a consequence enabling low-price generic drugs to inundate the market. Many brand name pharmaceutical manufacturers have contended with intensifying competition from generic manufacturers, cutting into revenue growth. Until the next the five years till 2022, investments in research and development that generate a high return will occur as many pharmaceutical manufacturers strengthen their drug pipeline with orphan drugs.
Recently, process engineers have shown inclination to single-use, modular and continuous manufacturing technologies to improve efficiency and minimize scale-up and technology transfer challenges in the pharmaceutical industry. Lot of these innovations are driven by updates instigated by the FDA. The changes are inevitable; especially in the already aging biopharmaceutical sector of the industry, where original processes are often overly complex, and are characterized by inefficiency, and occasionally, inherent unpredictability.
Track 4:QC & QA: Quality control and Quality assurance:
With greater attention to the following elements this session will discuss the all the key lookouts essential for the development of a QA/QC system in pharmaceutical manufacturing process and Requires implementation in tracking inventory compilation:
An inventory agency responsible for coordinating QA/QC activities:
- A QA/QC plan;
- General QC procedures
- Source category-specific QC procedures.
- QA review procedures;
- Reporting, documentation, and archiving procedures.
Market research analysts predict that the global automated industrial quality control (QC) market will grow steadily at a CAGR of around 8% by 2021.
Track 5: Pharmaceutical Manufacturing and Pharmacoeconomics:
Pharmaceutical Manufacturing is the procedure of Pharmaceutical Drugs by pharmaceutical manufacturing companies. It is under constant pressure to fast-track innovation and increase the speed at which they introduce successful drugs to market. The days of big pharmaceutical companies owning many manufacturing plants are slowly ending, and every company, no matter what size, is looking for ways to lessen costs to survive in an increasingly tough and unpredictable economic climate.
Pharmacoeconomics is analysed as a part of decision processes at several stages of drug development and drug marketing. Greater challenges in decision-making coupled with improvements in the techniques of pharmacoeconomic research point to a greater role for pharmacoeconomics into the new millennium. This in turn will have consequences for companies in the pharmaceutical industry.
Track 6:Quality by design(QbD) approach
“The Quality cannot be tested into the product, but it should be built into it.” QbD can be attributed as a Scientific, risk-based, holistic and proactive approach to the development of a pharmaceutical product. It is more of a deliberate design effort from product conception through commercialization requiring full understanding of how product attributes and process would relate to product performance. In FDA’s Office of New Drug Quality Assessment (ONDQA), a new risk-based pharmaceutical quality assessment system (PQAS) was established based on the application of product and process understanding. The difference between QbD for NDA and ANDA products is most apparent at the first step of the process. For an NDA, the target product profile is under development while for the ANDA product the target product profile is well established by the labeling and clinical studies conducted to support the approval of the reference product.
Track 7: Pharmaceutical Packaging and Logistics:
Packaging is one of the largest industry sectors in the world, worth several billions. There are three major types of pharmaceutical packaging, which are: solid packaging, semi-solids packaging and liquid packaging. The pharmaceutical packaging is a very profitable industry and it is estimated that for 2022 it will reach USD. 8.24 billion. Pharmaceutical packaging represents a meagre percentage of this colossal market. The Global Logistics Market is expected to grow at a CAGR of 7.0% from 2016-2021 and CAGR of 5.6% from 2021-2027. By 2020, pharma cold-chain logistics will be worth $16.7 billion, and non-cold chain at $77.1 billion. The market was estimated at $14.84bn in 2016 and is expected to grow to $28.75bn in 2027.
Track 8: Clinical trials and Pharmacovigilance:
The global clinical trials market size was valued at USD 40.0 billion in 2016 and is expected to grow at a CAGR of 5.7% until 2025. The demand for biosimilars testing is expected to increase in the U.S. The interventional study design is the most prominent method of conducting clinical trials across with globe. According to statistics provided by the U.S. FDA, over 126,000 trials are currently being conducted by means of this method. Based on indication, the oncology segment is anticipated to witness the fastest growth. According to various sources, more than USD 38.0 billion is currently spent by the healthcare industry towards preclinical and clinical development of oncology therapy products. Hence, it is anticipated to grow at a lucrative CAGR and contribute over USD 15.0 billion towards the clinical trials market by 2025. MENA’s clinical trial revenues is expected to increase almost 10-fold over the next decade, which will build an annual market of about $1 billion.
Track 9: Pharmacogenomics and Pharmacoepidemiology:
Pharmacogenomics and Pharmacogenomics can assist the physicians achieve the target of personalized medicine. Personalized medicine will come to mean not just the right drug for the right individual, but the right drug for the specific infection disease influence a specific individual. It is a major challenge in current clinical practice, drug development, and drug regulation.
Pharmacoepidemiology is that the study of applied medication and effects in massive numbers of people. It provides an estimate of the beneficial drug effect and an estimate of adverse drug effect in a population. It is said to be a combination of Clinical Pharmacology and Epidemiology.
Track 10: Pharmacology and Toxicology:
The Pharmaceutical Industry is directly impacted by the research conducted with prescription drugs, vaccines, and OTC drugs being manufactured based on findings from the study of life sciences. Clinical trials are conducted to ensure that products being developed are tested on how well they work on individuals affected by the diseases or conditions they are created to treat.
Toxicology is the scientific study of adverse effects that occur in living organisms due to chemicals. It involves observing and reporting symptoms, mechanisms, detection and treatments of toxic substances, in particular relation to the poisoning of humans.
Track 11: Pharma Market Research:
Two powerful megatrends — dramatic deceleration in U.S. market growth and significant restructuring of the healthcare system — are at play in the U.S. pharmaceuticals industry. The U.S. pharmaceutical market is the world’s most important national market. Together with Canada and Mexico, it represents the largest continental pharma market worldwide. The United States alone holds over 45 percent of the global pharmaceutical market. In 2016, this share was valued around 446 billion U.S. dollars. The biopharmaceutical companies in the United States exported goods in the amount of some 31 billion U.S. dollars during 2016. In the United States, there are an estimated 100,000 OTC drug products marketed and sold in a variety of outlets, such as pharmacies and convenience stores. TH revenue for the OTC drugs also add to the pharma market growth. Top markets for pharmaceutical products continue to be developed countries in Western Europe, East Asia, and North America with high per capita spending on healthcare, growing elderly populations, and advanced regulatory systems. Though ranked lower, there are growing opportunities in developing countries like China as incomes and healthcare spending increases. The pharmaceutical sector has consistently been one of the most R&D intensive industries in the United States. The research-based industry generally allocates around 15 to 20 percent of revenues to R&D activities and invests over $50 billion on R&D annually.
Health economics is a tool to help us prioritise different and sometimes competing health care interventions for these fixed resources and, in doing so, health care is treated as a commodity like any other. Knowledge of health economics coupled with political insight is essential to understand resource allocation and expenditure in a modern health care system. Pharmacists, with their unique knowledge of medicine, are crucial in using pharmacoeconomic analysis to influence expenditure and distribution of resources on medicines. Design and management of pharmacoeconomic studies
- cost and burden of illness studies,
- drug utilization,
- budget impact analysis,
- modeling and simulations
are very important in today’s pharmaceutical market. Moreover pharmacoeconomic approach towards
- Management of Consensus Meetings (Advisory Boards, Delphi Panels, etc.)
- Re-analysis of trials
- Implementation and customization with local data of pharmacoeconomic models
have greatly aided the pharma industries.
Track 13:Active Pharmaceutical Ingredients:
The API market in the US is as expected growing at a CAGR of 8% over the period 2014-2022. The market research and analysis estimates that in terms of geographic regions, the Americas will be the major revenue contributor to the active pharmaceutical ingredients market throughout the next four years. The increasing demand for generic drugs from countries such as the US, the growth in aging population, and the presence of an organized R&D structure for drug development are some of the major factors that will drive the growth of the market in this region. The key vendors in this sector are Abbott Laboratories, Aurobindo Pharma, Mylan and Teva Pharmaceutical Industries.
Track 14: Drug discovery and NCEs:
Each year, CDER approves series of new drugs and biological products. Some of these products are often innovative new products that never before have been used in clinical practice. FDA’s classification of a drug as an “NME” for review purposes is distinct from FDA’s determination of whether a drug product is a “new chemical entity” or “NCE” within the meaning of the Federal Food, Drug, and Cosmetic Act. According to Medscape the ratio of researched drugs to eventually approved therapies at between 5,000-to-1 and 10,000-to-1. Nonetheless, if a drug manages to gain FDA approval, the cumulative direct and indirect expenses are huge. As per estimated study performed by a well-known university, the true cost to bring a drug to market was US$2.56 billion. This figure is inclusive of just shy of $1.4 billion in average out-of-pocket costs to the drug developer, as well as $1.16 billion in time costs, which are essentially the expected returns that investors forgo while a drug is in development.
Track 15: Regulatory Authority Compliance:
With more stringent compliance parameters set by the USFDA the quality assurance of pharmaceutical products is ensured. In addition to cGMP, GLP and GCP practices as per the US Federal law pharma products should comply with 21CFR regulations. Also on a regular basis the FDA organizes compliance programs for the pharma industries in program areas like Biologics (CBER), Bioresearch Monitoring (BIMO), Devices/Radiological Health (CDRH), Drugs (CDER), Food and Cosmetics (CFSAN), Veterinary Medicine (CVM). The results of regulatory audits NAI – No Action Indication, VAI – Voluntary Actions Indicated, OAI – Official FDA Action Indicated are also critical to decide by auditors in regards to compliance matters and call for several challenges to overcome by the manufacturers.
Track 16: Novel Drug Delivery Systems, Biomarkers & Drug Targets:
The main advantage of Novel Drug Delivery System is providing the drug to the target site in a controlled manner for enhanced efficacy with less side effects. It follows two approaches one is spatial drug delivery which involves targeting a drug to a particular organ or tissue and another one is temporal drug delivery. In this the rate of the drug delivery to the target tissue is controlled by various techniques.
Track 17:Formulations and NNDS:
The drug delivery technology market is expected to reach USD 1,504.7 Billion by 2020 from USD 1,048.1 Billion in 2016, growing at a CAGR of 7.5% from 2015 to 2020. The global market for Novel Drug Delivery Systems (NDDS) is expected to hit USD 320 billion by the year 2021. With the rising number of patients suffering from different types of cancer, favorable reimbursement scenario in US, improved and advanced healthcare infrastructure have driven North America to become the leading regional segment in the global NDDS in cancer therapy market. Among the NDDS technologies Embolization of Particles, Selective Internal Radiation Therapy (SIRT), Holmium-based Microspheres, Liquid Embolics and Nanoparticles are gaining importance from market point of view.
Track 18:Analytical strategies for pharmaceutical products:
Chromatography and MS are extensively used in the formulation development stage of generic drug products. The ICH Q3B guidelines address the reporting threshold, identification threshold, and qualification threshold for impurities in the drug product. Chromatography (LC) and Gas Chromatography (GC) are extensively used to analyze stable compounds. However, for labile compounds require derivatization prior to LC or GC analysis. For certain selected analyses, Liquid Chromatography- Ultraviolet Detection (LC-UVD) and Gas Chromatography-Flame Ionization Detection (GC-FID) techniques may be deemed suitable. However, Liquid Chromatography-Mass Spectrometry (LC-MS) and Gas Chromatography-Mass Spectrometry (GC-MS) are usually the techniques of choice when higher specificity and sensitivity are required. Gas Chromatography-Electron Capture Detection (GC-ECD) techniques are commonly used for halogenated PGIs/GIs to enhance sensitivity and selectivity. Occasionally, some spectroscopic techniques like Nuclear Magnetic Resonance (NMR), light scattering, and Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) are used in analyzing PGIs/GIs.
Track 19: Clinical Pharmacy and Pharmacy Practices:
Clinical pharmacy and pharmacy practice play important roles in clinical trials and clinical research. In recent years pharmacist role in clinical research and patient care has improved significantly and they play active role in designing and modifying drug therapies. This session focuses on the recent advancements in clinical research and pharmacy practice.
Track 20: Bioequivalence and Bioavailability:
The main advantage of Novel Drug Delivery System is providing the drug to the target site in a controlled manner for enhanced efficacy with less side effects. It follows two approaches one is spatial drug delivery which involves targeting a drug to a particular organ or tissue and another one is temporal drug delivery. In this the rate of the drug delivery to the target tissue is controlled by various techniques.
Pharmaceutical Conference | Pharma Meeting | Pharmacology Symposium | Pharma Sciences Seminar | Pharmacoepidemiology Event | Pharmacogenomics Science Forum | Drug Delivery congress
Market Analysis
Pharmaceutical Excipients:
The global pharmaceutical excipients market is projected to reach USD 8.1 Billion in 2021 at a CAGR of 6.1% in the forecast period 2016 to 2024. The rising demand for new drug delivery systems, greater understanding of the functional benefits of excipients, growing pharmaceutical industry, and patent expiries of several blockbuster drugs are positively impacting the overall growth of the market.
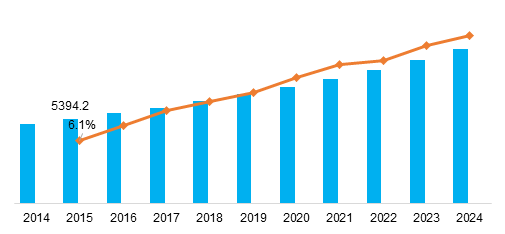
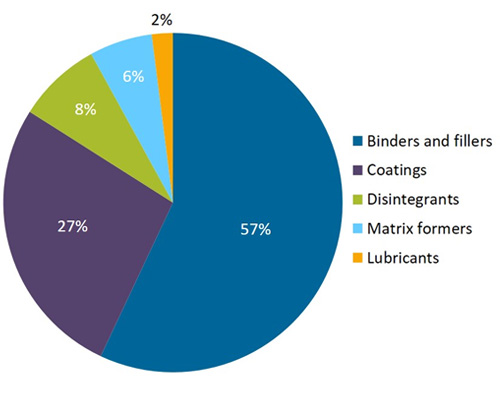
Global Pharmaceutical excipients Market, by Region
-
North America
- U.S.
- Canada
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Rest of Europe (RoE)
- China
- Japan
- India
- Rest of APAC
- Latin America
- Middle East & Africa
- Asia-Pacific (APAC)
- Rest of the World (RoW)
Major Marketing Associations around the Globe
- International Institute of Marketing Professionals (IIMP®)
- Marketing Power (American Marketing Association -AMA)
- American Branding Association (ABC)
- Canadian Marketing Association (CMA)
- Marketing Education Association (MEA)-USA
- Association for Consumer Research (ACR)
- The Asia-Pacific Professional Services Marketing Association (APSMA)
- Association of Internet Marketing and Sales (AIMS)-Canada
To Collaborate Scientific Professionals around the World
Conference Date May 16-17, 2024
For Sponsors & Exhibitors
Speaker Opportunity
Useful Links
Past Conference Report
Supported By
All accepted abstracts will be published in respective Conference Series International Journals.
Abstracts will be provided with Digital Object Identifier by





